|
(Clicking
on the thumbnail images will launch a new window and a larger version
of the thumbnail.) |
| Last updated: June 13, 2011. |
Odobenus rosmarus (divergens)
Order: Pinnipedia
Family: Odobenidae
1) General Zoological Data
Walruses are large, gregarious animals with both sexes having tusks. Males are larger (1,500 kg) than females (900 kg) and occur in Northern parts of Atlantic and Pacific oceans. Mating is said to take place in January/February and is followed by a 4-5 months delay in implantation (Nowak, 1999). Birth occurs 10-11 months thereafter, with the neonate weighing 50-63 kg. Several subspecies are nominated: Odobenus rosmarus rosmarus for the Atlantic animal and O. rosmarus divergens for the Pacific species; others are less well defined. The name (Odobenus) derives from the Greek for tooth and walking, as the animals have been observed to pull themselves over ice with their modified canines (tusks); and rosmarus derives from the Scandinavian denomination of the animal (Gotch, 1979). Longevity is 30 years according to Nishiwaki (1972). From his extensive study of pinniped chromosomes, Arnason (1974b) suggested a monophyletic origin of Pinnipedia. Different points of view were discussed by Duffield (1972). In their wide-ranging study of carnivore genomes, O'Brien et al. (1999) deduced that pinnipeds split from other carnivores approximately 30 MYA. Phylogenetic relations among the pinnipeds were strongly supported by the mitochondrial rRNA study of Ledje & Arnason (1996). Andersen et al. (1998) studied the population structure of Atlantic walruses with mtDNA fragments and found one animal with a Pacific walrus haplotype, thus proving a connection between the two populations.
 |
Adult male Pacific walrus. |
2)
General Gestational Data
The total length of gestation is 15-16 months (including the period of delayed
implantation) with singletons the rule. This delay of implantation is about
4-5 months long. There is one record of a set of twins, but the article
was not available to me (Nikulin, 1954), and Hayssen et al. (1993) make
reference to rare other twin gestations in Russian observations. Hybrids
with other species are unknown. General data on pinniped reproduction, including
walrus, may be found in the chapter by Costa & Crocker (1998).
3)
Implantation
There are no publications of implantation, and Mossman (1987) described
only one zonary placenta that he had observed superficially; he found
it to be zonary.
4)
General Characterization of the Placenta
The placenta of this term pregnancy was kindly made available by Dr. Judy
St. Leger of Sea World, San Diego. It comes from a multigravida that had
gained 800 lbs. during gestation and that was initially very protective
of the newborn calf, but eventually the newborn had to be hand-raised.
The placenta had remained in sea water for over a day but was cooled and
thus in an excellent macroscopic condition. There was minimal autolysis,
thus histology was also satisfactory but inadequate for electronmicroscopy.
Ordinarily, the placenta is eaten by gulls, according to Hayssen et al.
(1993).
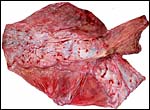
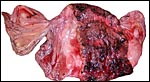
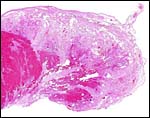
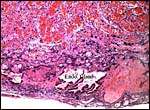
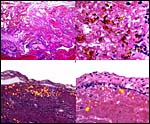
This is a typical zonary placenta weighing 5,900 g and measuring 85 cm in greatest length. The "ring" of placental tissue was 25 cm wide. It was complete and the circle was 100 cm in circumference. The villous tissue was 2-2.3 cm thick and darkly congested. At the sides there was the characteristic, irregular, brownish old hematoma, the so-called hemophagous organ. The maternal surface had much fresh clot attached and was irregular on its yellowish surface. There were no subdivisions of the placental tissues. The fetal surface was smooth although a focus of white, nodular squamous metaplasia of amnion was found (5x2 cm). The umbilical cord was mostly absent and less than 10 cm remained.
Another placenta was kindly made available by Dr. Carlos Rodriguez of the New York Aquarium. It weighed 4,980 g, had a similar zonary shape and the same ‘hemophagous’ peripheral region.
A third placenta, coming from a stillborn calf, was made available by Diana Procter (Vallejo, CA). It weighed 6,700 g, its cord was 36 cm long, 4.5 cm in circumference and 2.6 cm in diameter.
5)
Details of fetal/maternal barrier
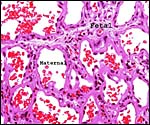
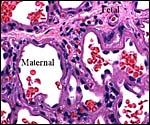
The walrus placenta, like that of other pinnipeds, is labyrinthine and much
resembles in its structure that of other carnivores, like the cat. The marginal
"hematoma" is also brown, rather than green as in the dog. I did
not find regions of active bleeding nor was there the central "hematoma"
described by others for seal placentas. There are, however, different regions
that can be identified grossly, but better microscopically, when inspecting
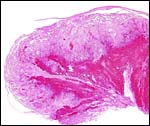
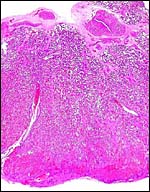
the cross-sectioned organ. The largest portion is the labyrinth which, in
my material, was very much congested. On top of the labyrinth of course
is the chorioallantoic mesenchyme (the chorionic plate with fetal vessels).
The base of the delivered placenta had some of the so-called "spongy
layer" attached but it lacked most of the maternal endometrial glands
that otherwise make up much of this zone. The base had debris, small bits
of endometrial glands surrounded by syncytium, fibrin and fresh blood. Although
decidua exists, little comes away with the delivered organ. At the base
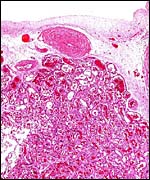
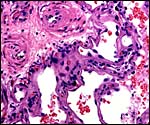
of the labyrinth there are irregular collections of syncytiotrophoblast,
the much darker cells in the photographs shown below. The deep red color
of the labyrinth of the walrus placenta shown here is from the presence
of enormously dilated maternal blood channels. They are lined by endothelium
and covered on the outside by trophoblast. This is thus an endotheliochorial
organ.
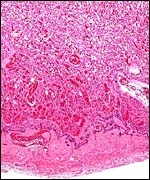
The trophoblast at the base is mostly syncytial in nature and the quite inapparent cytotrophoblast in the labyrinth appears to give rise to the syncytium. The syncytium is superficially invasive into the endometrium, destroys some of the stroma and opens some glands, but it does not grow into the maternal vascular lumens or into their walls.
The other placentas available all had the same microscopic appearance. The colored material at the edges was crystalline (square crystals) and iron stain negative.
6)
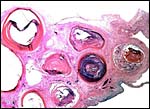
Umbilical cord
The normal length of the umbilical cord is unknown until the other two specimens were available. This specimen lacked
most of its cord and one vein had already split off from its cross section
near the placental surface. Normally it probably contains two arteries,
two veins and a large allantoic duct. Numerous small blood vessels are also
present that become more numerous near the placental surface where they
spread over the allantoic sac. There were no nodules on the remains of this
cord's amnionic surface. The umbilical cord of the New York specimen was between 45 and 50 cm long and that from Vallejo measured 36 cm in length, 4.5 cm in circumference and was 2.6 cm wide.
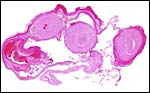 |
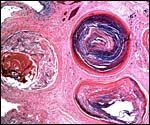
Cross section of cord immediately prior to its insertion on the placental surface - one vein has already departed. The central empty area is the beginning of the allantoic sac. |
7)
Uteroplacental circulation
This has not been described and could not be observed in this placenta other
than the visualization of endometrial arteries entering the labyrinth. They
are large channels and send blood to the undersurface of the chorion whence
the blood is returned through the labyrinthine channels.
8)
Extraplacental membranes
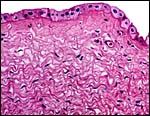
The amnion is lined by a single layer of cuboidal epithelium and has one
larger area of squamous inclusions. It contains no blood vessels. The
large allantois has a diffuse capillary vasculature and is lined by cuboidal
to columnar epithelium. Yolk sac remnants were not found in this term
placenta.
9)
Trophoblast external to barrier
The trophoblastic invasion of the decidua is very superficial and, in the
absence of a uterus with implanted placenta, no statement can be made about
deeper trophoblast invasion. This is absent, however, in other pinnipeds
and, since the larger endometrial vessels are not invaded (see pictures
above), it is unlikely that deeper penetration of trophoblast occurs in
walrus.
10)
Endometrium
Walruses have a bicornuate uterus. The glands of the endometrium were
partially destroyed in this term placenta but their bases remained. They
did not contain any obvious secretion. True decidual transformation cannot
be seen in the endometrial stroma.
11)
Various features
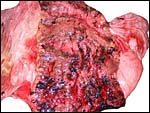
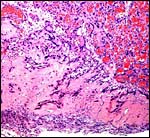
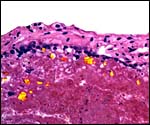
As many other carnivora, at the margin of this zonary placenta one finds
the "hemophagous zone", a border composed of degenerating villi,
fibrin and remnants of probable hematomas. In contrast to the dog, it
is not green but brown/yellow and laminated, much like that seen in the
cat, as was also remarked by Amoroso (1961) for related pinnipeds. Yellow
pigment occurs here and a few other areas of the placenta which has frequently
a crystalline form. The crystals are highly birefractory in polarized
light (unlike hemosiderin) and have a box-like shape. They do not stain
for iron with Prussian blue stains. Beneath the chorion the more columnar
trophoblast contains many crystals.
12)
Endocrinology
I am not aware that any publications on the endocrine performance of pregnant
walruses exist. A general survey of the reproductive physiology, including
some general aspects of endocrine performance of pinnipeds, is found in
Costa & Crocker (1998).
13)
Genetics
Pinnipedia have either 32 or 34 chromosomes; the Walruses have 32 chromosomes
with one metacentric pair having typical secondary constrictions (Fay
et al., 1967; Arnason, 1974a; Hsu & Benirschke, 1977; Arnason, 1977;
Arnason, 1981). The monophyletic origin of pinnipeds was affirmed by Arnason's
study (1974b). The NF of pinnipeds is 64. A characteristic secondary constriction
of a pair metacentrics and the very small Y chromosome are typical.
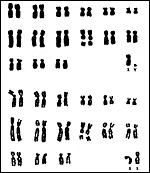 |
Chromosomes of male and female walruses (Courtesy U. Arnason). |
14)
Immunology
I know of no publications on the immunology of walruses.
15)
Pathological features
Various surveys for antibodies have been conducted in walruses and other
pinnipeds. Calle et al. (2002) found no antibodies against Brucella
sp. in the Pacific walrus, 3 of 40 had antibodies against Leptospira
interrogans, but phocine distemper was not detected. A variety of
other antibodies existed (against calicivirus, vesicular exanthema, and
various subtypes of influenza A). Nielsen et al. (2000) detected antibodies
against morbillivirus in 50 of Atlantic walruses and, in 2001, identified
antibodies to Brucella but they were unable to identify the organism.
Although Zarnke et al. (1997) identified antibodies to phocid herpesviruses,
they were of the opinion that herpes infection was not a cause of mortality.
Antibodies against caliciviruses were detected in Pacific walruses by
Barlough et al. (1986) who had also earlier identified the virus.
Trichinella infection is common in walrus and it is a source of infection
to the Inuits (Proulx et al., 2002). Brucellosis of sea mammals was studied
by Foster et al. (2002) who separated the responsible organism of terrestrial
animals from those in sea mammals (Brucella cetaceae & B.
pinnipedia). A listing of parasites found in walruses was provided
by Dailey & Brownell (1972).
Myocardial infarction and widespread atherosclerosis was the cause of
death in a 25 year-old walrus reported by Gruber et al. (2002).
Cornell et al. (1975) described an unusual complication of neonatal life,
the persistence of a tight fibrous band expanding from the dome of the
bladder to the umbilicus that prevented complete emptying of the bladder.
The about one month-old, 69 kg walrus had been hand-reared and developed
pyelonephritis and hydronephrosis from this complication and died. It
also had ascites and intestinal torsion.
16)
Physiologic data
No physiological studies on placenta or uterine perfusion have been undertaken
on walrus implantations. There is, however, an excellent consideration
of the development of the (probably similar) endotheliochorial placentation
in mink (Pfarrer et al., 1999). It described the development of the maternal
sinusoids from endometrial capillaries through the invasive action of
the syncytiotrophoblast in three developmental stages. Histologic studies
and corrosion casts were employed and these authors provided an excellent
overview of the features of placental perfusion in endotheliochorial placentas.
17)
Other resources
To the best of my knowledge, the only cell lines available are those of
Dr. U. Anderson at the University of Lund, Sweden.
18)
Other remarks - What additional Information is needed?
No implantational study has been undertaken and the length of the umbilical
cord needs to be ascertained. Endocrine studies are completely lacking.
Acknowledgement
The animal photograph in this chapter was kindly supplied by Dr. Sam Ridgway.
I very much appreciate the help of Dr. Judy St. Leger of Sea World, San
Diego for allowing me to study this placenta. Dr. Carlos Rodriguez, New York Wildlife Conservation Society kindly made the second placenta available. I also wish to acknowledge
the constant help of the histopathologists at UCSD Medical Center in preparing
the slides.
References
Amoroso, E.C.: Placentation. Chapter 15, pp.127-311, In, Marshall's Physiology
of Reproduction, V.II, A.S. Parkes, ed. Second Edition. Little, Brown
& Co. Boston, 1961.
Andersen, L.W., Born, E.W., Gjertz, I., Wiig, O., Holm, L.E. and Bendixen, C.: Population structure and gene flow of the Atlantic walrus (Odobenus rosmarus rosmarus) in the eastern Arctic based on mitochondrial DNA and microsatellite variation. Mol. Evol. 7:1323-1336, 1998.
Arnason, U.: Comparative chromosome studies in Pinnipedia. Hereditas 76:179-225, 1974a.
Arnason, U.: Phylogeny and speciation in Pinnipedia and Cetacea - a cytogenetic study. Thesis, Lund, 1974b.
Arnason, U.: The relationship between the four principal pinniped karyotypes. Hereditas 87:227-242, 1977.
Arnason, U.: Localization of nucleolar organizing regions in pinniped karyotypes. Hereditas 94:29-34, 1981.
Barlough, J.E., Berry, E.S., Skilling, D.E., Smith, A.W. and Fay, F.H.: Antibodies to marine caliciviruses in the Pacific walrus (Odobenus rosmarus divergens Illiger) J. Wildl. Dis. 22:165-168, 1986.
Calle, P.P., Seagars, D.J., McClave, C., Senne, D., House, C. and House, J.A.: Viral and bacterial serology of free-ranging Pacific walrus. J. Wildl. Dis. 38:93-100, 2002.
Cornell, L.H., Golden, B.J. and Osborn, K.G.: Pseudopersistent urachus in a baby walrus. J. Amer. Vet. Med. Ass. 167:548-549, 1975.
Costa, D.P. and Crocker, D.E.: Seals. Pp. 313-321, in Encyclopedia of Reproduction, Vol. 4, E. Knobil and J.D. Neill, eds. Academic Press, San Diego, 1998.
Dailey, M.D. and Brownell, R.L., Jr.: A checklist of marine mammal parasites. Chapter 9, pp. 528-589, in Mammals of the Sea. Biology and Medicine. S.H. Ridgway, ed. Charles C. Thomas, Springfield, Illinois, 1972.
Duffield Kulu, D.: Evolution and cytogenetics. Chapter 8, pp. 503-527, in Mammals of the Sea. Biology and Medicine. S.H. Ridgway, ed. Charles C. Thomas, Springfield, Illinois, 1972.
Fay, F.H., Rausch, V.R. and Feltz, E.T.: Cytogenetic comparison of some pinnipeds (Mammalia: Eutheria). Canad. J. Zool. 45:773-778, 1967.
Foster, G., MacMillan, A.P., Godfroid, J., Howie, F., Ross, H.M., Cloeckaert, A., Reid, R.J., Brew, S. and Patterson, I.A.: A review of Brucella sp. infection of sea mammals with particular emphasis on isolates from Scotland. Vet. Microbiol. 90:563-580, 2002.
Gotch, A.F.: Mammals - Their Latin Names Explained. Blandford Press, Poole, Dorset, 1979.
Gruber, A.D., Peters, M., Knieriem, A. and Wohlsein, P.: Atherosclerosis with multifocal myocardial infarction in a Pacific walrus (Odobenus rosmarus divergens Illiger). J. Zoo Wildl. Med. 33:139-144, 2002.
Hayssen,
V., van Tienhoven, A. and van Tienhoven, A.: Asdell's Patterns of Mammalian
Reproduction: a Compendium of Species-specific Data. Comstock/Cornell
University Press, Ithaca, 1993.
Hsu, T.C. and Benirschke, K.: An Atlas of Mammalian Chromosomes. Vol.
10, Folio 495, 1977.
Ledje, C. and Arnason, U.: Phylogenetic relationships within caniform carnivores based on analyses of the mitochondrial 12S rRNA gene. J. Mol. Evol. 43:641-649, 1006.
Mossman, H.W.: Vertebrate Fetal Membranes. MacMillan, Houndmills, 1987.
Nielsen, O., Stewart, R.E., Measures, L., Duignan, P. and House, C.: A morbillivirus antibody survey of Atlantic walrus, narwhal and beluga in Canada. J. Wildl. Dis. 36:508-517, 2000.
Nielsen, O., Stewart, R.E., Nielsen, K., Measures, L. and Duignan, P.: Serologic survey of Brucella spp. antibodies in some marine mammals of North America. J. Wildl. Dis. 37:89-100, 2001.
Nikulin, P.G.: On twinning in Chukchi walruses. Pacific Research Institute of Fisheries and Oceanography (TNIRO). Izvestia 39:353, 1954.
Nishiwaki, M.: General Biology. Pp. 162-164, in: Mammals of the Sea. Biology and Medicine. S.H. Ridgway, ed. Charles C. Thomas, Springfield, Illinois, 1972.
Nowak, R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press, Baltimore, 1999.
O'Brien, S.J., Menotti-Raymond, M., Murphy, W.J., Nash, W.G., Wienberg, J., Stanyon, R., Copeland, N.G., Jenkins, N.A., Womack, J.E. and Graves, J.A.: The promise of comparative genomics in mammals. Science 286:458-481, 1999.
Pfarrer, C., Winther, H., Leiser, R. and Dantzer, V.: The development of the endotheliochorial mink placenta: light microscopy and scanning electron microscopical morphometry of maternal vascular casts. Anat. Embryol. 199:63-74, 1999.
Proulx, J.F., MacLean, J.D., Gyorkos, T.W., Leclair, D., Richter, A.K., Serbir, B., Forbes, L. and Gajadhar, A.A.: Novel prevention program for trichinellosis in inuit communities. Clin. Infect. Dis. 34:1508-1514, 2002.
Zarnke, R.L., Harder, T.C., Vos, H.W., VerHof, J.M. and Oosterhaus, A.D.: Serologic survey for phocid herpesvirus-1 and -2 in marine mammals from Alaska and Russia. J. Wildl. Dis. 33:459-465, 1997.