|
(Clicking
on the thumbnail images below will launch a new window and a larger
version of the thumbnail.)
|
| Last updated: June 7, 2004. |
Equus asinus somalicus
(Equus africanus somaliensis)
Order: Perissodactyla
Family: Equidae
1) General Zoological Data
The Somali wild ass was once widely distributed over Northern Africa but is now either extinct in Africa or is very rare. Two subspecies of African wild asses (Somali and Nubian) are commonly recognized, the Nubian wild ass being somewhat smaller. Some 70+ of these wild asses are kept in many zoological gardens. Pitra et al. (1995) indicated that the captive population ultimately had derived from 3.6 founders and that it is thus significantly inbred. They also did some initial studies to ascertain the genetic diversity in the captive stock. In contrast to the domestic burro (the presumed derivative of the Nubian wild ass by domestication 6,000 years ago), Somali Wild Asses are pale, yellow. The typical Somali Wild Ass may have a faint stripe across the shoulder and also possesses delicate stripes on the legs; it has a short, standing mane. More details of these animals are found in Krumbiegel (1958), and Groves (1986) presented an excellent, broad overview of the African asses. He also reviewed the disagreement that exists concerning the notion that the domestic animal comes from the Nubian wild ass. The real facts are still outstanding. No doubt, much interbreeding of these wild asses with domestic donkeys, both in the wild and in captivity, further complicates the picture. Maximum life span is over 22 years in captivity.
 |
Somali wild ass mare and foal at San Diego Zoo's Wild Animal Park. |
 |
Somali wild ass stallion at San Diego's Wild Animal Park. |
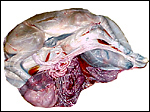
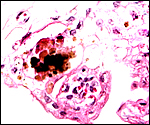
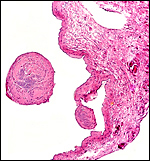
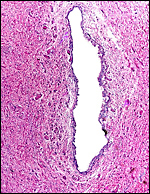
One placenta of this species was obtained from a mare that died with sepsis as a result of colonic obstruction by a huge, stone-hard fecalith. The female fetus was slightly autolyzed and weighed 15,700 g; it had a CR length of 80 cm. It was normally developed. On opening the uterus, the placenta was found to have been mostly detached from the uterine wall, but without there being any intervening blood. The brown placental surface had a nearly uniform cover of microvilli.
Another placenta came from a term gestation and weighed 2,180 g; it had a 32 cm long umbilical cord with three blood vessels. No macroscopic picture is available of that specimen. A third placenta in August, 2003 weighed 1,925 g, measured 52 cm in greatest diameter and had a 49 cm umbilical cord with four vessels.
Singletons are the norm and are born after an approximately 380 day gestation period (11 months). They weigh around 25-30 kg. Adults weigh around 250 kg.
3) Implantation
There are no data on early placentation in the Somali Wild Ass, while this has been described extensively in the domestic horse (see that chapter).
4)
General Characterization of the Placenta
The placenta of all equidae is superficially similar, nearly identical.
That of the Somali Wild Ass has not been described but it is extremely
similar to that of the domestic donkey. A most detailed description of
equine placentation is presented in the chapter on the domestic horse
to which the reader is referred. Another chapter of this web site discusses
the Kiang placenta, and other information is found in the chapter on the
Grevy zebra. This Somali Wild Ass placenta is also an epithelio-chorial,
diffuse placenta with large allantoic sac and without invasion of the
uterus. The single exception, the invasion at the chorionic girdle, could
not be studied in this mature organ. At this site, trophoblastic invasion
occurs over a smaller area than that of the horse (see chapter on horse).
This region produces the eCG of equine placentas, less in donkey than
in horse.
This is a rather diffuse epithelio-chorial placenta with simple villi that abut the endometrial surface. There is no trophoblastic invasion of the uterus, except perhaps at the chorionic girdle. That region was not identified in this term organ, as one may see it in the delivered horse placenta (see chapter on domestic horse). It is likely, however, that a girdle with invasion and eCG production exists in this species. If younger specimens become available, that aspect needs verification.
The trophoblast is cuboidal and uninuclear, binucleate cells were not found. Under the chorioallantoic plate, the trophoblast is more cylindrical, especially in between the villi (the "areolar" regions). As in the domestic horse and many ungulate placentas, this cylindrical trophoblast often has yellow pigment granules within the cytoplasm. Iron stains (Prussian blue) are negative. The nature of this pigment is uncertain. It is not associated with hemorrhages, as is suggested by the term "haemophagous organ" that is extensively discussed in sheep placentation. I do not believe this pigment to derive from hemoglobin breakdown. Alternatively, I suggest that it may represent melanin.
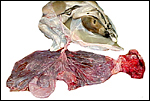
The umbilical cord inserted at the cranial part of the uterus, had three blood vessels and a large allantoic duct. The amnionic portion measured 28 cm, the allantoic portion was 10 cm long. Another cord, from a term delivered placenta, measured 32 cm in length and was otherwise identical. The cord is partitioned into a somewhat longer amnionic portion, where its vessels are contained in Wharton's jelly and associated with smaller vessels and the allantoic duct, and the allantoic section. The latter commences at the site of insertion of the allanto-amnion and corresponds to the opening of the allantoic duct into the allantois. Here, the cord vessels are separated and only loosely associated and then diverge over the allantoic surface. In the amnionic portion of the cord it is covered with plaques of sqamous metaplasia. The vascular split of the allantoic vessels, as is usual in equines, is well shown in the gross photographs above. Aside from the two large arteries and one vein, numerous small blood vessels are found in the cord. Some, but only a minority, associate with the allantoic duct.
Two additional placentas at term with 32 and 49 cm long umbilical cords had three and four blood vessels, respectively.
The vasculature of the horse placenta has been beautifully described by Steven (1982), and subsequently by Wooding and Flint (1994). The reader is referred to the chapter on the domestic horse for a much more detailed discussion of this aspect.
8)
Extraplacental membranes
The amnion is composed of a thin layer of connective tissue with simple
epithelium. It covers the portion of cord closest to the fetus and has
small areas of squamous metaplasia near the fetal region of the cord.
It is not vascularized and is almost completely surrounded by allantoic
fluid and its outside is thus covered by allantoic sac membrane, to form
the "allantoamnion". The allantoic membrane carries large numbers
of blood vessels and it contains fetal urine. Large, flat brown hippomanes
were present in this sac. On cross sectioning they are laminated with
darker centers. I did not find remnants of yolk sac. If it anything like
the horse, then small remnants may be present at the fetal insertion of
the cord. There is no decidua.
There is no trophoblastic infiltration of the endometrium, except in the small "girdle" during early gestation. This has not been seen in the wild ass but is best studied in the domestic horse and burro. That infiltration is temporary and is the site of eCG production.
10)
Endometrium
The equine placentation is not associated with typical decidualization.
Steven (1982) has traced vasculature of endometrium during gestation.
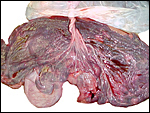
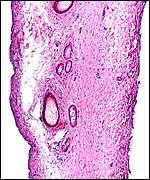
Following delivery and contraction of the uterus the appearance of the
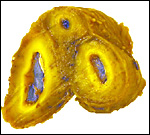
endometrium is that as shown in the next photograph. The superficial portions,
now thickened by contraction, are the glands with which the villi had
intersected. The unmodified uterine glands are at the base, near the myometrium.
 |
Uterus after placental separation. |
No other specific features are worth mentioning that have not been referred to in other portions of this chapter.
12)
Endocrinology
Butt et al. (1994) have summarized the findings of gonadotropin production
in equine gestations. They referred to the study by Aggarwal et al. (1980)
of their partial purification of a chorionic gonadotropin from horse and
donkey. These glycoproteins were similar in size but the donkey gonadotropin
had 31% (vs. 45% in horse) carbohydrate content. It had LH-activity in
bioassays.
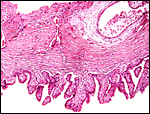
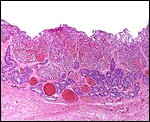
The fetal ovaries measured 4x2x2 cm and had their usual dark brown appearance
that is so characteristic of equidae. The size and color are the result
of stimulation of a large mass of interstitial cells. While one may assume
that this relates to the presence of eCG in equine gestations, the amount
of that hormone in the circulation has long disappeared when the ovaries
(and testes) are stimulated. Besides, it is not found in the fetal circulation.
Thus, the stimulus for the interstitial cell development is still unknown.
Many endocrine studies have been undertaken in domestic donkeys, but none
are published for the Somali Wild Ass. The interested reader is referred
to the chapter on the domestic horse for further details.
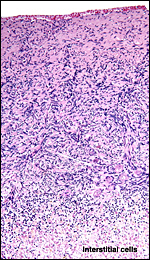 |
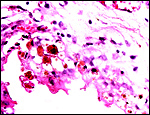
Section of the fetal ovarian cortex showing the dense connective tissue, a small group of oocytes (white arrows) and the edge of the large mass of brown interstitial cells at bottom. |
All donkeys, including the wild asses, have normally 62 chromosomes (Ryder et al., 1978). As a consequence, hybrids between African donkeys and domestic donkeys are fertile, as are hybrids between the Nubian (E. asinus africanus) and the Somali Wild Asses (E. asinus somalicus). Gray (1972) lists a hybrid with a kulan at Rome, but does not discuss its fertility.
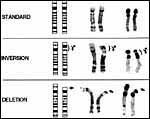
A much more extensive study of the chromosomes, including banding patterns, was done by Houck et al. (1998). It involves a pedigree of Somali Wild Asses at the San Diego Zoo and revealed novel findings. Animals with 2n=62, 63, and 64 chromosomes were identified. In addition, some animals had a heritable pericentric inversion of chromosome # 2, and animals with 63 chromosomes were fertile. It was presumed that fission in a heterochromatic region was the mechanism of this chromosomal variety.
To my knowledge there have been no immunological studies on Somali Wild Asses.
15)
Pathological features
This animal died from obstruction of the colon by a very large fecalith
that had led to sepsis. Impaction (sand) has been reported by Griner (1983)
in burros. He did not have Somali Wild Asses for necropsy. Ascaridiasis
is another common problem with captive equids.
16)
Physiologic data
No physiological studies have been undertaken on this severely endangered
species. It is likely, however, that findings would be similar in most
respects to those summarized in the chapter on the domestic horse.
17)
Other resources
Cell strains of frozen fibroblasts are available from CRES, the research
arm of the Zoological Society of San Diego by contacting Dr. Oliver Ryder
at oryder@ucsd.edu.
18) Other remarks - What additional Information is needed?
Acknowledgement
The animal photographs in this chapter come from the Zoological Society
of San Diego. I appreciate also very much the help of the pathologists
at the San Diego Zoo.
References
Aggarwal, P.B., Farmer, S.W., Papkoff, H. and Stewart, F.: Purification
and characterization of donkey chorionic gonadotrophin. J. Endocrinol.
85:449-455, 1980.
Butt, W.R., Chard, T. and Iles, R.K.: Hormones of the placenta: hCG and hPL. Pp. 461-534 of Volume 3, Part one, in Marshall's Physiology of Reproduction, fourth edition, G.E. Lamming, ed. Chapman & Hall, London, 1994
Gray, A.P.: Mammalian Hybrids. A Check-list with Bibliography. 2nd edition. Commonwealth Agricultural Bureaux Farnham Royal, Slough, England, 1972.
Griner, L.A.: Pathology of Zoo Animals. Zoological Society of San Diego, San Diego, California, 1983.
Groves, C.P.: The taxonomy, distribution, and adaptations of recent equids. Pp. 11-65, in Equids in the Ancient World. Meadow, R.H. and Uerpmann, H.-P. eds. Dr. Ludwig Reichert Verlag, Wiesbaden, 1986.,
Houck, M.L., Kumamoto, A.T., Cabrera, R.M. and Benirschke, K.: Chromosomal rearrangements in a Somali wild ass pedigree, Equus africanus somaliensis (Perissodactyla, Equidae). Cytogenet. Cell Genet. 80:117-122, 1998.
Krumbiegel, I.: Einhufer. A. Ziemsen Verlag, Wittenberg, 1958.
Pitra, C., Streich, W.J., Reinsch, A., Fickel, J. and Mann, W.: Die Population des Somali-Wildesels (Equus africanus somalicus Sclater) in menschlicher Obhut: Demographische und genetische Aspekte. Zool. Garten 65:245-257, 1995.
Ryder, O.A., Epel, N.C. and Benirschke, K.: Chromosome banding studies of the equidae. Cytogenet. Cell Genet. 20:323-350, 1978.
Steven,
D.H.: Placentation in the mare. J. Reprod. Fertil. Suppl. 31:41-55, 1982.
Wooding, F.B.P. and Flint, A.P.F.: Placentation. Chapter 4 (pp.233-460)
in, G.E. Lamming, ed. Marshall's Physiology of Reproduction, 4th ed. Vol.
3, Part 1. Chapman & Hall, London, 1994.