| (Clicking
on the thumbnail images will launch a new window and a larger version
of the thumbnail.) |
| Last updated: May 22, 2004. |
Capricornis crispus
By Hiroaki Soma, Tokyo & K. Benirschke
Order: Artiodactyla
Family: Bovidae
1) General Zoological Data
There are two species of serow, Capriconis sumatrensis and Capriconis crispus. The latter has two subspecies, the Japanese and Formosan serows. The Japanese serow is a mountainous, endangered species in Japan that is being conserved at the Japanese Serow Center on Gozaisho Mountain.
The presumed evolution of the Rupicaprinae has been traced from fossils and bone structures by Dolan (1963) and it is also graphically depicted below. Speciation is presumed to have proceeded via intermediate species from chamois-like ancestors (Soma & Kada, 1984) to the Rocky Mountain Goat as its latest derivative. More recently, the phylogenetic relationships have been studied by examination of mtDNA and, especially, nuclear DNA genes of proteins. Thus, Chikuni et al. (1995) undertook a major effort to study nucleotide substitutions of a wide variety of Bovidae for the kappa-casein and cytochrome b gene sequences and compared their results with findings from other molecular studies. In their view, this essential protein and its genes provided better resolving power than did mtDNA evolution. They suggested a cattle/pig split to have occurred 60 MYA, Cattle/sheep & goat 20 MYA, and Sheep/goat 5 MYA. Their results also place the chamois as being more ancestral, whence derived Japanese serow, and then the other two serow species.
Adult serows weigh between 50 and 100 kg and both sexes have short curved horns (Nowak, 1999) whose crystalline components were studied in detail by Hashiguchi & Hashimoto (1995). Further general details on Japanese serows have been provided by Benirschke & Soma (1972 published (in Japanese); and by Benirschke (1973; Soma & Kada).
 |
Japanese Serow at Gozaisho Mountain, Japan. |
 |
Japanese Serow with young at San Diego Zoo's Wild Animal Park. |
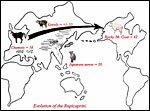 |
Presumed chromosomal evolution of Rupicaprini. |
According to Nowak (1999), the gestation is 7 months long and usually singletons are born. The recorded longevity is 18 years 7 months in captivity (Jones, 1993). General details on reproduction and neonatal growth were published by Sugimura et al. (1981) in a paper that was not accessible.
3) Implantation
No studies of early stages of placentation are known to us.
4) General Characterization of the Placenta
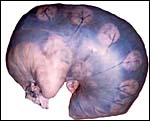
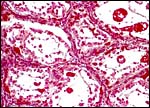
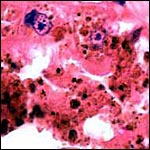
KB had one placenta available for dissection. It came from a neonatal death occurring at San Diego Zoo's Wild Animal Park and its gestation was at term. The placenta was slightly autolyzed and weighed 470 g. This placenta had 60 cotyledons measuring up to 4 cm in diameter in the main uterine horn, but the cotyledons were as small as 0.5 cm in the secondary horn. There was only a 1 cm umbilical cord attached to the membranes. Its surface was covered by thin amnionic epithelium.
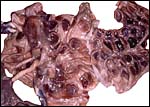
In addition, Hiroaki Soma of Tokyo had available the material from two other animals collected at the Serow Center of Gozaisho Mountain, and the Yokohama Zoo, Japan. Both of these specimens came from stillbirths at 212 and 213 days gestation. The first placenta was attached to a uterus and had 91 cotyledons with maximal diameter of 4 cm. Uterus and placenta weighed 675 g, the male fetus was 760 g, and the cord measured 13 cm in length and possessed four vessels. The other case was a stillbirth with a fetus 41 cm in CH length having a 15 cm cord (4 vessels) and 72 placental cotyledons measuring up to 6.5 cm in diameter. Pictures of both will be shown next. Since this original description a placenta from a term, surviving infant at San Diego Zoo's Wild Animal Park became available. It weighed 110 g and had only 14 large (4 cm) flat cotyledons. A 12 cm umbilical cord with four vessels and allantoic duct was present.
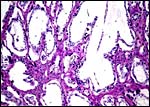
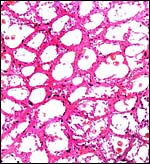
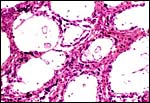
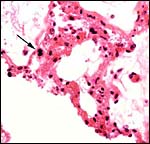
This is an epitheliochorial placentation with tall cylindrical trophoblast covering the villi and interspersed with numerous binucleated trophoblast cells. The villi in the slightly autolyzed placenta from San Diego are much more edematous than is probably normal. Moreover, it is not so prominent in the other two placentas shown. In the trophoblast under the chorionic plate one finds relatively small quantities of yellow pigment.
6)
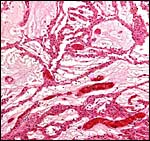
Umbilical cord
Clearly, the 1 cm length of umbilical cord that was received in the first
specimen is an incorrect length, but the real length of the umbilical cord
was unknown until the Japanese specimens became available. The piece received
initially was attached to the placenta and had already split into its several
large surface vessels and the allantoic sac was opened. Thus, the early
finding of only three large blood vessels was incorrect. The two Japanese
specimens had four blood vessels (and allantoic duct), were 13 and 15 cm
long, and had a left spiral.
The new placenta had a 12 cm cord as well that was similarly structured.
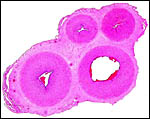 |
Cross section of umbilical cord of Japanese serow. |
7) Uteroplacental circulation
No studies are known to us.
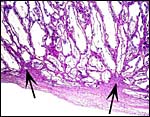
The allantoic sac is large and lined by cylindrical epithelium and has a large number of blood vessels. The thin amnion contains no vessels. No remnants of yolk sac were found.
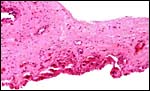 |
Intercotyledonary membrane with trophoblast below. |
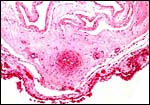 |
Amnion above, allantois below. |
There is no trophoblastic infiltration of endometrium or uterus. In view of the general similarity to ungulates with similar placentas, however, such infiltration seemed highly unlikely anyway.
10) Endometrium
Whether there is endometrial decidualization is unknown. Very little typical
decidua was present in the term uterus of a dead dam.
11) Various features
A subplacenta is not present.
12) Endocrinology
We know of no endocrine studies in this animal.
The Japanese serow has 50 chromosomes (Benirschke et al., 1972; Ito et al., 1972; Soma & Kada, 1984). The Sumatran serow has 2=48 (Soma et al., 1987), and the Formosan subspecies of Capricornis crispus (Capricornis crispus swinhoei) is identical to that of the Japanese animal (Soma & Kada, 1984). Presumably, Robertsonian fusion mechanisms were operative in the speciation, as all species have a "NF" of 60.
 |
Karyotype of male Japanese Serow. |
Sugimura et al. (1990) described the composition of lymph nodes in a variety of animals, including the serow, but we are not aware of any other immunological studies, other than surveys. Thus, Inoshima et al. (1999; 2001) surveyed Japanese serows and found a 20% immune response to parapox virus.
15) Pathological features
A macerated fetus was found by Yamamoto et al. (1994) among "2,138
pregnant Japanese serows". The fetus appears to have been retained,
as all placentomes had disappeared and there was an inflammatory reaction.
Parapoxvirus infection of the interdigital glands was found in a wild
serow by Suzuki et al. (1997a). Numerous parasites have been identified
in serows, thus: Inoue (1989) found four species of Eimeria that
were non-infectious for goats; Gongylonema was identified by
Chakraborty (1994); Inoue & Imura (1991) identified Eimeria gozaishoensis
in a Formosan serow, and Uni et al. (2001, 2002) isolated several Cercopithifilaria
species from Japanese serows. A wild serow with dermatitis was found to
suffer from infection by Chorioptes bovis, apparently a new host
for this mite (Shibata et al., 2003). Onchocerca was found to
cause lesions of the ligaments to the olecranon, with chronic tendonitis
(Suzuki et al., 1997b).
16) Physiologic data
The serow in Japan is protected and at the same time it is pursued because
of its raiding crops. Therefore, Deguchi et al. (2001) conducted a study
to ascertain food preferences. They found that the animals prefer crop
rather than wild plants and inferred that this relates to the higher protein
content of crops.
A large number of anatomical studies have been performed on this rare
animal. Yamamoto et al. (1998) and Kitamura et al. (2003) described the
microscopic findings of the forestomach anatomy. Atoji et al. (1996) found
significant differences in the structure of the infraorbital glands of
Formosan and Japanese serows. The masseter muscle structure was described
by Suzuki (1989), and the interdigital gland of a Formosan serow was described
by Atoji et al. (1995) and compared with that of a Japanese serow (Atoji
et al., 1987). Bronchial tree and lungs were examined by Nakakuki (1986).
Atoji et al. (1987a) studied the brachial plexus and later, the lumbosacral
plexus of Japanese serows (1987b). The same authors also examined the
histology of preputial glands (Atoji et al., 1989). Takahashi et al. (1984)
determined the lipid composition of red cell membranes in serows, and
Kita et al. (1995) described mammary glands and teats.
17)
Other resources
Cell strains of fibroblasts are available from CRES
at the San Diego Zoo by contacting Dr. Oliver Ryder at oryder@ucsd.edu.
18) Other remarks - What additional
Information is needed?
No early stages of implantation have been described and would be desirable
to obtain. Endocrine studies during pregnancy are needed.
Acknowledgement
The first animal photograph in this chapter comes from Mr. Takeuchi at
the Japan Serow Center, Gozaisho Mountain, Japan.
References
Atoji, Y., Hori, Y., Sugimura, M. and Suzuki, Y.: Lectin histochemical
study on the
infraorbital gland of the Japanese serow (Capricornis crispus).
Acta Morphol. Neerl. Scand. 25:201-213, 1987.
Atoji, Y., Suzuki, Y. and Sugimura, M.:
The brachial plexus of the Japanese serow
(Capricornis crispus). Anat. Anz. 163:25-32, 1987a.
Atoji, Y., Suzuki, Y. and Sugimura, M.: The lumbosacral plexus of the Japanese serow, Capricornis crispus. Anat. Anz. 164:213-217, 1987b.
Atoji, Y., Suzuki, Y. and Sugimura, M.: Lectin histochemistry of the interdigital gland in the Japanese serow (Capricornis crispus) J. Anat. 161:159-170, 1988.
Atoji, Y., Suzuki, Y. and Sugimura, M.:
The preputial gland of the Japanese serow
Capricornis crispus: ultrastructure and lectin histochemistry.
Acta Anat. 134:245-252, 1989.
Atoji, Y., Yamamoto, Y. and Suzuki, Y.:
Morphology of the interdigital glands of a
Formosan serow (Capricornis crispus swinhoei). J. Vet.
Med. Sci. 57:963-964, 1995.
Atoji, Y., Yamamoto, Y. and Suzuki, Y.: Infraorbital glands of a male Formosan serow (Capricornis crispus swinhoei). Eur. J. Morphol. 34:87-94, 1996.
Benirschke, K.: The Japanese serow. ZooNooz 46:14?17, 1973.
Benirschke, K., Soma, H. and Ito, T.:
The chromosomes of the Japanese serow,
Capricornis crispus (Temminck). Japan Acad. 48:608?612, 1972.
Benirschke, K. and Soma, H.: The Japanese
serow. Life Sciences 10:1?4, 1972
(Japanese).
Chakraborty, A.: Occurrence and pathology
of Gongylonema infection in captive
herbivores. Vet. Parasitol. 52:163-167.
Chikuni, K., Mori, Y., Tabata, T., Saito, M., Monma, M. and Kosugiyama, M.: Molecular phylogeny based on the ?-casein and cytochrome b sequences in the mammalian suborder Ruminantia. J. Mol. Evol. 41:859-866, 1995.
Deguchi, Y., Sato, S. and Sugawara, K.:
Relationship between some chemical
components of herbage, dietary preference and fresh herbage intake rate
by the Japanese serow. Appl. Anim. Behav. Sci. 73:69-79, 2001.
Dolan, J.: Beitrag zur systematischen Gliederung des Tribus Rupicaprini Simpson. Z. Zool. Syst. Evolutionsforsch. 1:311-407, 1963.
Hashiguchi, K. and Hashimoto, K.: The
mineralization of crystalline inorganic
components in Japanese serow horn. Okajimas Folia Anat. Jpn. 72:235-243,
1995.
Inoshima, Y., Shimizu, S., Minamoto, N., Hirai, K. and Sentsui, H.: Use of protein AG in an enzyme-linked immunosorbent assay for screening for antibodies against parapoxvirus in wild animals in Japan. Clin. Diagn. Lab. Immunol. 6:388-391, 1999.
Inoshima, Y., Yamamoto, Y., Takahashi, T., Shino, M., Katsumi, A., Shimizu, S. and Sentsui, H.: Serological survey of parapoxvirus infection in wild ruminants in Japan in 1996-1999. Epidemiol. Infect. 126:153-156, 2001.
Inoue, I.: Eimeria Capricornis n .sp.,
E. nihonis n. sp., E. naganoensis n. sp. and E.
kamoshika n. sp. (Protozoa; Eimeridae) from the Japanese serow, Capricornis
crispus. Nippon Juigaku Zasshi. 51:163-168, 1989.
Inoue, I. and Imura, M.: Eimeria gozaishoensis
n. sp. from the Formosan serow
(Capricornis crispus swinhoei). J. Wildl. Dis. 27:214-216,
1991.
Ito, T., Soma, H. and Benirschke, K.: The chromosomes complement of the Japanese serow, Capricornis crispus crispus. Mammalian Chromosomes Newsletter 13:12?13, 1972.
Jones, M.L.: Longevity of ungulates in captivity. Intern. Zoo Yearb. 32:159-169, 1993.
Kita, I., Miura, S., Kojima, Y. and Tiba, T.: Macroscopic observations of mammary glands and teats of Japanese serows, Capricornis crispus, with special reference to past gestation. J. Vet. Med. Sci. 57:447-451, 1995.
Kitamura, N., Yoshiki, A., Sasaki, M., Baltazar, E.T., Honda, E., Yamamoto, Y., Agungpriyono, S. and Yamada, J.: Immunohistochemical evaluation of the muscularis mucosae in the ruminant forestomach. Anat. Histol. Embryol. 32:175-178, 2003.
Nakakuki, S.: The bronchial tree and blood vessels of the Japanese serow lung. Anat. Anz. 161:61-68, 1986.
Nowak, R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press, Baltimore, 1999.
Shibata, A., Yachimori, S., Morita, T., Kanda, E., Ike, K. and Imai, S.: Chorioptic mange in a wild Japanese serow. J. Wildl. Dis. 39:437-440, 2003.
Soma, H.: and Kada, H.: Evolutionary pathway
of chromosomes of the Capricornis. Chapter 11, pp. 109-118, in, One Medicine,
O.A. Ryder and M.L. Byrd, eds. Springer-Verlag, Berlin, 1984.
Soma, H. Kada, H. Matayoshi, K. Evolutionary pathways of karyotypes of
the tribe Rupicaprini. In Soma, H. (ed) The Biology and management of
Capricornis and Related Mountain Antelopes. Croom Helm, London, 62-71,
1987.
Sugimura, M., Suzuki, Y., Kamiya, S. and Fukita, T.: Reproduction and prenatal growth in the wild Japanese serow, Capricornis crispus. Nippon Juigaku Zasshi. 43:553-555, 1981.
Sugimura, M., Shirogane, D., Atoji, Y., Suzuki, Y., Ohshima, K., Kon, Y. and Hashimoto, Y.: A comparative study on S-100 protein-immunoreactive cells in lymph nodes. Nippon Juigaku Zasshi 52:1015-1021, 1990.
Suzuki, T.: The lamination of the masseter muscle in the Japanese serow (Capricornis crispus). Okajimas Folia Anat. Jpn. 65:381-389, 1989.
Suzuki, Y., Komatsu, T., Yamamoto, Y. and Atoji, Y.: Pathology of interdigital glands in a wild Japanese serow (Capricornis crispus) infected with parapoxvirus. J. Vet. Med. Sci. 59:1063-1065, 1997a.
Suzuki, Y., Uni, S., Komatsu, T., Yamamoto, Y. and Atoji, Y.: Olecranon lesions caused by Onchocerca skrjabini in wild Japanese serows (Capricornis crispus). J. Vet. Med. Sci. 59:387-390, 1997b.
Takahashi, M., Imai, A., Nakashima, S. and Nozawa, Y.: Phospholipid and fatty acid composition of erythrocyte membrane from wild Japanese serow (Capricornis crispus). Comp. Biochem. Physiol. B 77:369-371, 1984.
Uni, S., Suzuki, Y., Baba, M., Mitani, N., Takaoka, H., Katsumi, A. and Bain, O.: Coexistence of five Cercopithifilaria species in the Japanese rupicaprine bovid, Capricornis crispus. Parasite 8:197-213, 2001.
Uni, S., Bain, O., Takaoka, H., Katsumi, A., Fujita, H. and Suzuki, Y.: Diversification of Cercopithifilaria species (Nematoda: Filaroidea) in Japanese wild ruminants with description of two new species. Parasite 9:293-304, 2002.
Yamamoto, Y., Atoji, Y., Kita, I. and Suzuki, Y.: Maceration of fetus in the Japanese serow (Capricornis crispus). J. Vet. Med. Sci. 56:165-166, 1994.
Yamamoto, Y., Atoji, Y., Agungpriyono, S... and Suzuki, Y.: Morphological study of the forestomach of the Japanese serow (Capricornis crispus). Anat. Histol. Embryol. 27:73-81, 1998.