| (Clicking
on the thumbnail images will launch a new window and a larger version
of the thumbnail.) |
| Last updated: October 26, 2003 |
Hippotragus niger
Order: Artiodactyla
Family: Bovidae
1) General Zoological Data
There are three species in the genus Hippotragus: H. leucophaeus (the blue buck - now extinct since 1800), H. equinus (the roan antelope), and H. niger (the sable antelope). Several subspecies have been described of sable and of roan antelopes (H. equinus). H. n. variani is the rare giant sable antelope (Nowak, 1999). other subspecies have been described by Groves (1983). The name derives from hippos (Greek - horse) and tragos (male goat). H.F. Varian described the giant sable of Angola (H. n. variani) in 1913 (Gotch, 1979). Indeed, Blaine (1922) considered the Angolan subspecies to be sufficiently different as to assign it species rank. The "blue buck ("Blaubock") found detailed description in the monograph by Erna Mohr (1967).
Sable antelopes are said to "share a close evolutionary relationship and together form a sister clade to the more primitive Caprinae" (Matthee & Davis, 2001). Hassanin & Douzery (1999) suggested that they arose during one of the two major explosive radiatio ns occurring between 12 and 15.3 MYA.
Jones (1993) lists longevity for two sable antelope subspecies (12-22years), with the South African animal (H. n. niger) having the record of 22 years and 3 months. This is currently not the case. Wilson & Hirst (1977) presented a detailed monograph on ecology of South African Roan and Sable antelopes. (See also the chapter on Roan antelope).
 |
Sable antelope at San Diego Zoo. |
The length of gestation is reported to be 268-280 days in roan antelopes and 240-281 in sables (Nowak, 1999; Mentis, 1972). Single offspring are the norm. Neonates weigh 16 kg (Ferrell et al., 2001). The attached placenta shown in this chapter comes from a gestation that ended in maternal death. The male fetus weighed 1,125 g and had a 30 cm CR length. The breeding behavior of sable antelope has been described in detail by Buechner et al. (1974).
3) Implantation
No specific data on early implantation have been published. Mossman & Mossman (1969) examined 58 pregnant uteri and all were implanted in the right uterine horn, irrespective of ovulation site (27 right/28 left). They likened it to the situation existing in Adenota kob. The sex ratio was 1:1. Moreover, when they examined 9 virgin uteri, the right uterine horn was longer than the left.
4) General Characterization of the Placenta
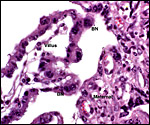
This is a polycotyledonary placenta with epitheliochorial relationship. Hradecky (1983) found one sable antelope placenta to have 185 cotyledons measuring 3x2 cm, and another with only 109 cotyledons, arranged in six rows. The first placenta we examined had nearly 100 oval cotyledons. Another one had 50+ cotyledons and weighed 820 g. The nature of the sable antelope cotyledon was described by Hradecky et al. (1988a) as composed of villi 4 mm long. The maternal septa were relatively thick and "did not fully match the shapes of the contained villi" (there was more infolding of villous surfaces).
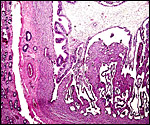
 |
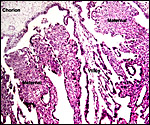
Young, implanted cotyledon ("placentome") with uterus below. |
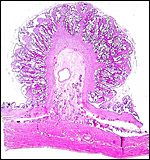
 |
This sable placenta was detached from the endometrium (below) after fetal and then maternal demise. |
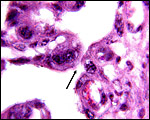
The trophoblast is cuboidal with microvillous surface and closely applied to the endometrial epithelium of the crypts which also have a microvillous surface. There is the usual number of typical, large binucleate trophoblastic cells, probably also engaged in the production of placental lactogen, as is the case in other ungulates. The villous connective tissue stroma is delicate and contains a small number of macrophages (Hofbauer cells). The villi are sparsely branched. Beneath the chorionic plate and in between cotyledons, the trophoblast is much more cylindrical but in none of my specimens contains any pigment. Moreover, none of the specimens had degeneration of the septal tips or hemorrhages.
The umbilical cord has four large blood vessels, an allantoic duct and a moderate number of small vessels. Its surface is remarkable because of the numerous "callosities", areas of squamous metaplasia with rather marked keratin production. It is less than 20 cm long but good measurements have not been available. There are no spirals. The second placenta had a 10 cm long umbilical cord.
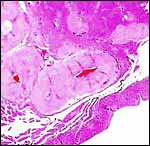
 |
Umbilical cord with numerous foci of surface keratin accumulations and allantoic duct at center bottom. |
This has not been described.
8)
Extraplacental membranes
The amnion is as remarkable as the surface of the umbilical cord with
its large number of keratinized plaques. Hradecky (1984) measured the
amounts of allantoic and amnionic fluid in one gestation with a 31 cm
fetus. There was twice as much allantoic than amnionic fluid, and its
composition more nearly resembled fetal urine.
There is no infiltration of trophoblast into the endometrial stroma or blood vessels.
10)
Endometrium
The uterus is bicornuate and implantation is always in the right horn,
which is longer than that on the left (Mossman & Mossman, 1969). There
are typical caruncles, not all of which may be employed during gestation.
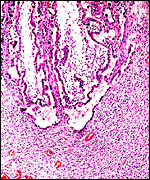
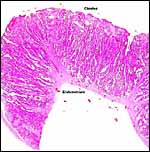
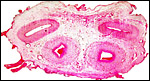
 |
Unused caruncle from the uterus of a pregnant sable antelope. The thick endo/myometrial vessels suggest that it had formerly been used as a placentome. |
There is no subplacenta and endometrial secretion in intercotyledonary areolae has been alleged to exist through electron microscopic observations. This was also suggested by the observations made by Hradecky et al. (1988b) on implanted placentas.
12)
Endocrinology
I have not been able to find any information on endocrine studies of sable
antelope gestations. Fetal/neonatal gonads show no signs of being stimulated;
the fetal adrenals are small. Except for the corpus luteum of pregnancy
and rare ovarian cystic follicles, the maternal ovaries appear unstimulated
in gestation.
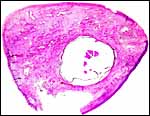 |
Cross section of one maternal ovary whose uterus contained fetus and placenta. There is one cystic follicle, otherwise only scars of former ovulations. |
The sable antelope possesses 60 chromosomes, all acrocentrics (Wurster & Benirschke, 1968). The Y chromosome is minute, compared with the other elements. Claro et al. (1993) have made more detailed studies of this species' chromosomes, also finding 60 chromosomes but identifying the X as more similar to that of goat than cattle. To my knowledge, hybrids have not been reported. A more detailed banding study by Fordyce-Boyer et al. (1995) found subtle differences between roan and sable antelope chromosomes.
 |
Karyotypes of male and female sable antelopes (from Hsu & Benirschke, 1969). |
I know of no immunological studies on sable antelopes.
15)
Pathological features
Griner (1983) found neonatal mortality (and stillbirths) the most important
factors in his large series of autopsies at San Diego Zoo. Heuschele et
al. (1984) found neutralizing antibodies to the virus causing malignant
catarrhal fever in sable antelopes.
16)
Physiologic data
Numerous biomedical parameters were studied in 35 neonates by Ferrell
et al. (2001).
17) Other resources
There are numerous cell lines in the "Frozen Zoo" of CRES
at the San Diego Zoo that can be made available by contacting Dr. O. Ryder
at: oryder@ucsd.edu.
18)
Other remarks - What additional Information is needed?
There is no concrete information on the length of umbilical cords, no
electronmicroscopy has been done, and there are no data on early implantations
nor have endocrine parameters during pregnancy been published.
Acknowledgement
The animal photograph in this chapter comes from the Zoological Society
of San Diego. The best-preserved sections of placental tissue were kindly
donated by Dr. Petr Hradecky.
References
Blaine, G.: Notes on the zebras and some antelopes of Angola. Proc. Zool.
Soc. London # 22:317-339, 1922.
Buechner, H.K., Stroman, H.R. and Xanten, W.A.: Breeding behaviour of
Sable antelope Hippotragus niger in captivity. Intern. Zoo Yearb.
14:133-137, 1974.
Claro, F., Hayes, H. and Cribiu, E.P.: The R- and G-bands of the sable antelope (Hippotragus niger). J. Hered. 84:481-484, 1993.
Ferrell,
S.T., Radcliffe, R.W., Marsh, R., Thurman, C.B., Cartwright, C.M., de
Maar, T.W.J., Blumer, E.S., Spevak, E. and Osofsky, S.A.: Comparisons
among selected neonatal biomedical parameters of four species of semi-free
ranging hippotragini: Addax (Addax nasomaculatus), Scimitar-horned
oryx (Oryx dammah), Arabian oryx (Oryx leucoryx), and sable
antelope (Hippotragus niger). Zoo Biol. 20:47-54, 2001.
Fordyce-Boyer, R., Sanger, T., Loskutoff, N., Kumamoto, A.T., Johnston,
L. and Armstrong, D.: Comparative cytogenetic study of the Roan and Sable
antelope, Hippotragus equinus and Hippotragus niger. Applied
Cytogenet. 21:189-191, 1995.
Gotch, A.F.: Mammals - Their Latin Names Explained. Blandford Press, Poole, Dorset, 1979.
Griner,
L.A.: Pathology of Zoo Animals. Zoological Society of San Diego, San Diego,
California, 1983.
Groves, C.P.: A new subspecies of sable antelope, Hippotragus niger
(Harris 1838). Rev. Zool. Afr. 97:821-828, 1983.
Hassanin, A. and Douzery, E.J.: The tribal radiations of the family Bovidae (Artiodactyla) and the evolution of the mitochondrial cytochrome b gene. Mol. Phylogenet. Evol. 13:227-243, 1999.
Heuschele, W.P., Oosterhuis, J., Anderson, M.P., Swansen, M. and Fletcher, H.R.: Malignant catarrhal fever in wild ruminants. Chapter 25 (pp. 296-308) In, One Medicine; O.A. Ryder and M. L. Byrd, eds. Springer-Verlag, New York, 1984.
Hradecky, P.: Placental morphology in African antelopes and giraffes. Theriogenology 20:725-734, 1983.
Hradecky, P.: Composition of the fetal fluids in African antelopes. Theriogenology
Hradecky, P., Mossman, H.W. and Stott, G.G.: Comparative histology of antelope placentomes. Theriogenology 29:693-6729, 1988a.
Hradecky, P., Mossman, H.W. and Stott, G.G.: Comparative development of ruminant placentomes. Theriogenology 29:715-729, 1988b.
Hsu, T.C. and Benirschke, K.: An Atlas of Mammalian Chromosomes. 3: Folio 142, 1969.
Jones, M.L.: Longevity of ungulates in captivity. Int. Zoo Yearb. 32:159-169, 1993.
Matthee, C.A. and Davis, S.K.: Molecular insights into the evolution of the family Bovidae: A nuclear DNA perspective. Mol. Biol. Evol. 18:1220-1230, 2001.
Mentis, M.T.: A review of some life history features of the large herbivores of Africa. The Lammergeyer 16:1-89, 1972.
Mohr, E.: Der Blaubock. Hippotragus leucophaeus (Pallas, 1766). Eine Dokumentation Paul Parey, Hamburg, 1967.
Mossman, A.S. and Mossman, H.W.: Ovulation, implantation, and fetal sex ratio in impala. Science 137:869, 1962.
Nowak,
R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press,
Baltimore, 1999.
Wilson, D.E. and Hirst, S.M.: Ecology and factors limiting Roan andn Sable
antelope populations in South Africa. Wildlife Monograph #54 in, J. Wildl.
Management 41:1-111, 1977.
Wurster, D.H. and Benirschke, K.: Chromosome studies in the superfamily Bovoidea. Chromosoma 25:152-171, 1968.