| |
16)
Physiologic data
Since this animal is a browser, living in dense forest, the types of vegetation
consumed have been of interest. Gijzen's monograph (1959) provides a good
list of plants consumed. Rüedi et al. (1984) provided information on
the collection of semen from okapis. Rabb (1978) provided hematologic and
chemical data.
Lang (1956) described the maintenance of okapis at Epulu, the rapid "domestication"
of the animals, their ability to jump a 1.1 m high fences, and the adoption
of young by non-lactating females.
17)
Other resources
Many cell lines are available from CRES
at San Diego Zoo from its "Frozen Zoo" and may be accessed by
contacting Dr. O. Ryder (oryder@ucsd.edu).
18)
Other remarks - What additional Information is needed?
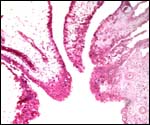
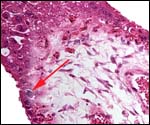
The reason for the pigmentary deposits in the neonatal endometrium is
obscure and needs an explanation. Are the amounts of placental lactogen
similar to the giraffe and are there placental/fetal gonadotropins? The
marked difference in the structure of fetal ovaries when compared with
the giraffe neonate needs an explanation. The collection of normative
data on okapis was emphasized earlier (Benirschke, 1978b).
Acknowledgement
I appreciate also very much the help of the pathologists at the San Diego
Zoo.
References
Anonymus: Okapi bibliography. Okapi 1-20, 1963.
Benirschke,
K.: General survey of okapi pathology. Acta Zool. Pathol. Antv. 71:63-78,
1978a.
Benirschke,
K.: Concluding remarks on the desirability of collecting normative data
on okapi. Acta Zool. Pathol. Antv. 71:131-134, 1978b.
Benirschke, K. and Hagey, L.R.: Should the okapi be listed as a giraffid species? Zool. Garten 76:197-198, 2006.
Benirschke,
K., Kumamoto, A.T., Cousin, E.F.H.M. and de Boer, L.E.M.: Further observations
on the chromosome of the okapi. (Okapia johnstoni) 25th Intern.
Sympos. Erkrank. Zootiere, Vienna, 1983. Akademie-Verlag, Berlin. Pp.
363-372, 1983.
Bois
de, H. and van Elsacker, L.: Bibliography of the okapi Okapia johnstoni
- "Another 20 years later". Acta Zool. Pathol. Antv. 80:65-84,
1988.
Bois
de, H., Dhont, A.A. and Puijenbroeck van, B.: Effects of inbreeding on
juvenile survival of the okapi Okapia johnstoni in captivity. Biol.
Conserv. 54:147-155, 1990.
Colbert,
E.H.: The relationships of the okapi. J. Mammal. 19:47-64, 1938.
Gallagher,
D.S., Davis, S.K., de Donato, M. Burzlaff, J.D., Womack, J.E., Taylor,
J.F. and Kumamoto, A.T.: A karyotypic analysis of the nilgai, Boselaphus
tragocamelus (Artiodactyla: Bovidae). Chromosome Res. 6:505-513, 1998.
Geurden,
L.M.G.: Les facteurs physio-pathologiques influençant l'acclimatation
de l'okapi. Bull. S.R. de Zoologie d'Anvers. # 3:1-21, 1953.
Gijzen,
A.: Notice sur la reproduction de l'okapi Okapia johnstoni (Sclater)
au Jardin Zoologique d'Anvers. Bull. S.R. de Zoologie d'Anvers. # 8:1-62,
1958.
Gijzen, A.: Das Okapi. (Neue Brehm-Bucherei). A. Ziemsen Verlag. Wittenberg
Lutherstadt, 1959.
Gijzen,
A.: Studbook of the okapi. Acta Zool. Pathol. Antv. 68:1-47, 1977. (continued
by C. Kruyfhooft).
Gijzen,
A. and Mortelmans, J.: Notice complémentaire sur l'okapi Okapia
johnstoni (Sclater), sur sa reproduction et ses maladies au Jardin
Zoologique d'Anvers. Bull. S. R. Zool. D'Anvers # 301-66, 1962.
Gijzen,
A. and Smet, S.: L'okapi en captivité depuis sa découverte
en 1900 (description par Sclater en 1901) jusqu'au 31 décembre
1970. Un pedigree pour l'okapi (Okapia johnstoni) (Sclater, 1901).
Chronique des okapis anversois. "Zoo". S.R. Zool. D'Anvers.
37:pp13, 1972.
Griner,
L.A.: Pathology of Zoo Animals. Zoological Society of San Diego, San Diego,
California, 1983.
Hamilton,
W.R.: Fossil giraffes from the Miocene of Africa and a revision of the
phylogeny of the Giraffoidea. Phil Trans. Roy. Soc. London B. 283(996):165-229,
1978.
Hösli,
P. and Lang, E.M.: A preliminary note on the chromosomes of the Giraffidae:
Giraffa camelopardalis and Okapi johnstoni. Mamm. Chromos.
Newsl. 11:109-110, 1070.
Hradecky,
P.: Placental morphology in African antelopes and giraffes. Theriogenology
20:725-734, 1983.
Hradecky,
P., Benirschke, K. and Stott, G.G.: Implications of the placental structure
compatibility for interspecies embryo transfer. Theriogenology 28:737-746,
1987.
Koulisher,
L.: Mammalian chromosomes. IX. The chromosomes of a female specimen of
Okapia johnstoni. Acta Zool. Pathol. Antv. 71:87-92, 1978.
Lang,
E.M.: Haltung und Brunst von Okapia in Epulu. Säugetierk.
Mitt. IV (2):49-52, 1956.
Lang,
E.M.: Einige Beobachtungen an Okapia johnstoni. Acta Tropica 13:254-258,
1956.
Loskutoff,
N.M., Ott, J.E. and Lasley, B.L.: Urinary steroid evaluations to monitor
ovarian function in exotic ungulates: I. Pregnanediol-3-glucoronide immunoreactivity
in the okapi (Okapia johnstoni). Zoo Biol. 1:45-53, 1982.
Loskutoff,
N.M., Kasman, L.H., Raphael, B.L., Ott-Joslin, J.E. and Lasley, B.L.:
Urinary steroid evaluations to monitor ovarian function in exotic ungulates:
IV. Estrogen metabolism in the okapi (Okapia johnstoni). Zoo Biol.
6:213-218, 1987.
Loskutoff,
N.M., Raphael, B.L., Dorn, C.G., Nemec, L.A., Calle, P.P., Petric, A.M.
and Kraemer, D.C.: Comparative reproductive traits of the okapi and giraffe:
implications for intraspecific and intergeneric embryo transfer. Acta
Zool. Pathol. Antv. 80:29-42, 1988.
Naaktgeboren,
C.: Notiz über einen trächtigen Uterus von Okapia johnstoni
(Sclater, 1901). Z. Säugetierk. 31:171-176, 1966.
Nouvel,
J.: Remarques sur la fonction genital et la naissance d'un okapi. Mammalia
22:107-111, 1958.
Nowak,
R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press,
Baltimore, 1999.
Petit,
P. and de Meurichy, W.: On the chromosomes of the okapi Okapia johnstoni.
Ann. Génét. 29:232-234, 1986.
Puijenbroeck
v., B.: Studbook of the okapi Okapia johnstoni (Sclater). Royal
Society of Antwerp, 1987.
Rabb,
G.B.: Birth, early behavior and clinical date on the okapi. Acta Zool.
Pathol. Antv. 71:93-105, 1978.
Raphael,
B.L.: Neonatal illness characterized by dermatitis, hyperthermina and
anemia in an okapi. Acta Zool. Pathol. Antv. 80:43-52, 1988.
Rüedi,
D., Küpfer, U., Matern, B., Klöppel, G. and Heldstab, A.: Semen
collection in the okapi. J. Zoo Med. 15:3-4, 1984.
Schwarzenberger,
F., Patzl, M., Francke, R., Ochs, A., Biter, R., Schaftenaar, W. and de
Meurichy, W.: Fecal progestagen evaluations to monitor the estrous cycle
and pregnancy in the okapi (Okapia johnstoni). Zoo Biol. 12:549-559,
1993.
Spinage,
C.A.: The Book of the Giraffe. Houghton Mifflin Company, Boston, 1968.
Symposium
on the okapi. Acta Zool. Pathol. 71:1-134, 1978.
Thenius,
E. and Hofer, H.: Stammesgeschichte der Säugetiere. Springer-Verlag,
Berlin, 1960.
Ulbrich,
F. and Schmitt, I.: Die chromosome von Okapia johnstoni. Acta Zool.
Pathol. Antv. 49:123-124, 1969.
Vandermander,
A.: Apercu succinct de nos connaissances actuelles au sujet de l'okapi
(Okapia johnstoni Scl.). Bull. S.R. de Zoologie d'Anvers #3: 22-46,
1953.
Vermeesch,
J.R., de Meurichy, W., van den Berghe, H., Marynen P. and Petit, P.: Differences
in the distribution and nature of the interstitial telomeric (TTAGGG)n
sequences in the chromosomes of the giraffidae, okapi (Okapia johnstoni),
and giraffe (Giraffa camelopardalis): evidence for ancestral telomeres
at the okapi polymorphic rob(4;26) fusion site. Cytogenet. Cell Genet.
72:310-315, 1996.
Zwart,
P., Gispen, R. and Peters, J.C.: Cowpox in okapis Okapia johnstoni
at Rotterdam zoo. Br. Vet. J. 127:20-24, 1971.
|