| (Clicking
on the thumbnail images will launch a new window and a larger version
of the thumbnail.) |
| Last updated: Sept 18, 2006. |
Oreotragus oreotragus
Order: Artiodactyla
Family: Bovidae
1) General Zoological Data
This widely dispersed African species (with eleven nominated subspecies Walther, 1968) weighs 8-18 kg in adulthood (Nowak, 1999). It behavior is characteristic and it is a strictly monogamous animal; only the males have short horns (10 cm). The animals have prominent dark preorbital glands below their eyes. Their maximum lifespan of 25 years was achieved by an Ethiopian specimen at Frankfurt , while usually their life ends around 15 years (Weigl, 2005). The animal's name derives from the Greek ( oros = mountain) and ( tragus = he- goat), while klip' is the Dutch for a rock, according to Gotch (1979). There is an active breeding colony at San Diego Zoo.
Wilson & Reeder (1993) include the klipspringer as members of the subfamily Antilopinae'; Thenius (1960) comprises them under Neotraginae and most authors admit that their taxonomic position is still somewhat controversial (Matthee & Davis, 2001). This is especially well summarized by Hernández Fernández & Vrba (2005). They referred to Rebholz & Harley (1999) who considered the tribe Neotragini as being sister species to the Antilopini on the basis of mtDNA studies. The cytochrome b study of Matthee & Robinson (1999) made the klipspringer a sister taxon of the Cephalophinae which is supported by those species all having similar numbers and structures of their chromosomes (see chapter on Yellow-backed duiker). Moreover, the research on pituitary and CNS structures by Haarmann (1975) and Oboussier (1979) have also suggested close affinities to Cephalophinae.
 |
Typical adult klipspringer at San Diego Zoo. |
2) General Gestational Data
Gestation is 214-225 days long according to Nowak (1999) with singletons being the norm and weighing around 1 kg.
3) Implantation
Early stages of placentation have not been reported and would be of great interest. In fact, I cannot find references to any placental study in this species.
4) General Characterization of the Placenta
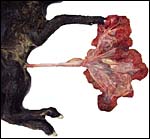
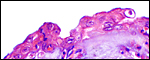
The specimen obtained comes from a delivery of a freshly male stillborn that was nearly totally black-skinned. The fetus weighed 1,100 g and had a crown-rump length of 35 cm. The neonate appeared mildly malnourished and the reasons for its fetal demise are not clear. The fetus and placenta were extracted by the veterinarian when the fetal head of the stillborn appeared and the labor had stopped.
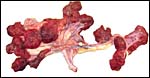
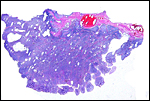
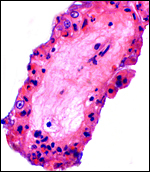
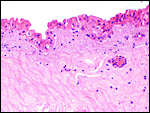
The placenta weighed 148 g and had 14 cotyledons that measured up to 4 cm in diameter. They were extremely mushy' from autolysis. No pathologic features were recognized macroscopically. Unfortunately, this placenta was relatively poorly preserved because of the fetal demise. Consequently, there is much edema in the villi. This is an oligocotyledonary villous placenta that contains relatively few cotyledons. Similar to the Cephalophinae, this placenta has a simple cuboidal trophoblastic cover of villi with a small number of binucleate cells. No pigmented subchorial trophoblast was present in this placenta either.
5) Details of fetal/maternal barrier
6) Umbilical cord
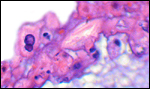
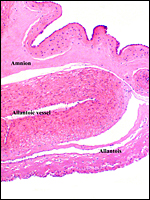
The cord measured 11 cm in length, was not spiraled and contained four vessels and a large allantoic duct. There was no squamous metaplasia in the cord's surface.
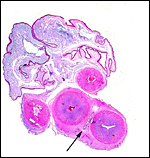 |
The umbilical cord has its allantoic duct (arrow) between the two arteries. |
7) Uteroplacental circulation
No such studies have been reported.
8) Extraplacental membranes
Amnion and allantoic sacs are present, but there is no remnant of yolk sac. The amnion has flat epithelium without caruncles. There was no decidua.
9) Trophoblast external to barrier
Although no such studies have been reported, it is unlikely that trophoblast invades endometrium or the uterus.
10) Endometrium
The uterus is bicornuate; decidua is not formed.
11) Various features
There is probably no subplacenta but an implanted placenta has not yet been described.
12) Endocrinology
No studies have been conducted in my review of the literature.
13) Genetics
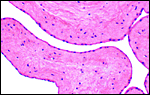
At CRES of the San Diego Zoo, numerous specimens of Klipspringer have been examined. All specimens of O. oreotragus stevensoni had 60 chromosomes, as yet unreported but individually identified (see O'Brien et al., 2006) the Y-chromosome is also acrocentric and is approximately 2/3 of the size of the X chromosome; it is shown as G-banded karyotype by O'Brien et al. (2006 p.635). At the beginning of this review, several studies are referred to by a variety of authors who have studied certain gene products in order to further delineate the evolution of this species and its relationship to other Bovidae. It should also be mentioned that the number of chromosomes and their structure are very similar to those of the yellow-back duiker (see that chapter) and thus support the closer relationship of the klipspringer to duikers. No hybrids have been reported (Gray, 1972).
 |
Karyotype of female klipspringer with 2n=60. All are acrocentrics. |
14) Immunology
I am not aware of any such studies.
15) Pathological features
Griner (1983) reported on the only klipspringer then in the collection at San Diego . The animal had liver cirrhosis and multiple hepatic staphylococcal abscesses. The origin for the infection was not ascertained.
16) Physiologic data
Raphael et al. (1991) have experimentally recovered ova and fertilized them.
17) Other resources
Frozen fibroblast cells lines are available from CRES at San Diego Zoo by contacting Dr. Oliver Ryder at oryder@ucsd.edu.
18) Other remarks What additional Information is needed?
Obviously, an implanted placenta and early stages of placentation are needed, in addition to further observations on the number and shape of cotyledons. As is true of other bovid placentas, the absence of pigmentation defies the notion that this haemophagous zone' is needed for iron supply to the fetus.
Acknowledgement
The animal photograph comes from the Zoological Society of San Diego.
References
Gotch, A.F.: Mammals Their Latin Names Explained. Blandford Press, Poole , Dorset , 1979.
Gray, A.P.: Mammalian Hybrids. A Check-list with Bibliography. 2 nd edition.
Commonwealth Agricultural Bureaux Farnham Royal, Slough , England , 1972.
Griner, L.A. : Pathology of Zoo Animals. Zoological Society of San Diego , San Diego , California, 1983.
Haarmann, K.: Morphological and histological study of neocortex of bovides (Antilopinae, Cephalophinae) and Tragulidae with comments on evolutionary development. J. Hirnforsch. 16:93-116, 1975.
Hernández Fernández , M. and Vrba, F.S.: A complete estimate of the phylogenetic relationships in Ruminantia: a dated species-level supertree of the extant ruminants. Biol. Rev. Camb. Philos. Soc. 80:269-302, 2005.
Matthee , C.A. and Davis , S.K.: Molecular insights into the evolution of the family Bovidae: A nuclear DNA perspective. Mol. Biol. Evol. 18:1220-1230, 2001.
Matthee , C.A. and Robinson, T.J.: Cytochrome b phylogeny of the family Bovidae: resolution within the alcelaphini, antilopini, neotragini, and tragelaphini. Mol. Phylogenet. Evol. 12:31-46, 1999.
Nowak, R.M.: Walker 's Mammals of the World. 6 th ed. The Johns Hopkins Press, Baltimore, 1999.
Oboussier, H.: Problems of body structure and size of the hypophysis. Acta anat. (Basel) 104:374-381, 1979.
O'Brien, S.J., Menninger, J.C. and Nash, W.G. eds. Atlas of Mammalian Chromosomes, Wiley & Sons, Hoboken , N.J. 2006.
Raphael, B.L., Loskutoff, N.M., huntress, S.L. and Kraemer, D.C., Postmortem recovery, in vitro maturation, and fertilization of Klipspringer (Oreotragus o reotragus) ovarian oocytes. J. Zoo Wild. Med. 22: 115-118, 1991.
Rebholz, W. and Harley, E.: Phylogenetic relationships in the bovid subfamily Antilopinae based on mitochondrial DNA sequences. Mol. Phylogenet. Evol. 12:87-94, 1999.
Thenius, E. and Hofer, H.: Stammesgeschichte der Säugetiere. Eine Uebersicht über Tatsachen und Probleme der Evolution der Säugetiere. Springer-Verlag , Berlin, 1960.
Walther, F: Ducker, Böckchen und Waldböcke. Chapter 12, pp. 342-367, in: Grzimek, B.: Grzimek's Tierleben. Enzyklopädie des Tierreiches. Kindler Verlag, Zurich , 1968, Vol. 13/4.
Weigl, R.: Longevity of Mammals in Captivity from the Living Collections of the World. E. Schweizertbart'sche Verlagsbuchhandlung (Nägele und Obermiller), Stuttgart, 2005.
Wilson , D.E. and Reeder, D.A.M.: Mammal Species of the World. A Taxonomic and Geographic Reference. 2 nd ed. Smithsonian Institution Press, Washington, DC, 1993.