|
(Clicking
on the thumbnail images below will launch a new window and a larger
version of the thumbnail.)
|
| Last updated: Aug 16, 2005 |
Nubian
Ibex
Capra ibex nubiani
Order:
Artiodactyla
Family: Bovidae, Caprinae
1)
General zoological data of species
There are perhaps eight "good" species of wild goats (Corbet,
1978), but there is much controversy attending the classification of this
genus (Nowak & Paradiso, 1983). The ibex is widely distributed, including
Africa, and it has a number of subspecies. They used to occur in groups
numbering dozens of animals, but they are now rare and are considered
to be threatened by extinction. The Nubian ibex is smaller than the Alpine
form and has scimitar-shaped horns. The one distinguishing feature is
the shape of their horns; their anterior surfaces are flat and broken
by prominent transverse ridges. Subsequent to writing this chapter, specimens
of Tur, Alpne ibex and Marhor became available. Please refer to the chapter
on Alpine ibex that summarizes these findings.
2)
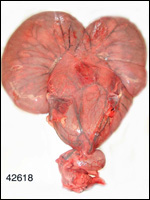
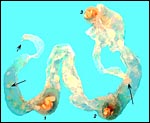
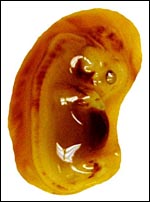
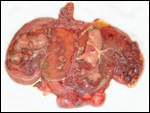
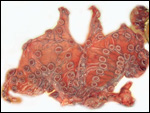
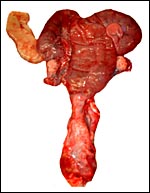
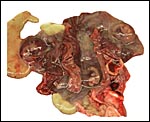
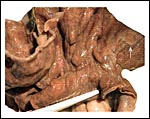
General gestational data
In addition to the Nubian ibex, the placenta of an West Caucasian Tur
(Capra caucasica), a related species, was available. Reference
to its placenta will be made below. The length of gestation in the Nubian
ibex is given as being between 147-180 days (Nowak & Paradiso, 1983;
Puschmann, 1989). The litter size is usually one, but twins and, very
rarely triplets, occur. The newborns weigh between 3.5 and 5 kg in Alpine
ibex but are smaller in Nubian ibex, 1.0-1.9 kg (our average weight recorded
in San Diego is 1.3 kg). The Nubian ibex is endangered. This was true
of the Alpine subspecies until conservations measures were taken to conserve
it.
The maternal weight is lower than the male weight of 60 kg; it was around
26 kg in the only specimen we had. The placental weight at term is unknown
to us. Breeding the ibex is seasonal and occurs in the summer. The length
of gestation is between 147-180 days (Nowak & Paradiso, 1983), with
twins occurring occasionally. Longevity is 22 years. Successful colonies
exist in the San Diego Zoo.