| |
9)
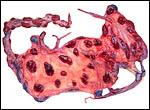
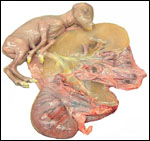
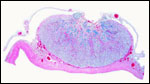
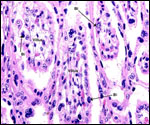
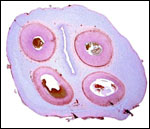
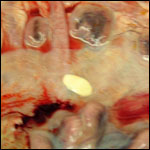
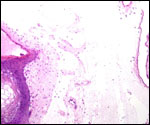
Trophoblast external to barrier
There is no extravillous trophoblast or invasion of the endometrium.
10)
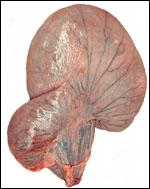
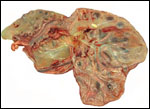
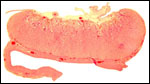
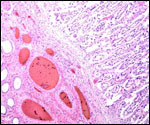
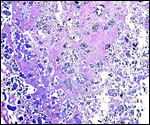
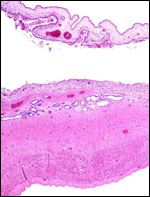
Endometrium
No true decidua is formed. The endometrial tissue becomes fibrous in appearance
beneath the cotyledon and under the intercotyledonary membrane. Small
numbers of lymphocytes are found within the stroma. Glands are present
only at the margin of cotyledons and in between the cotyledons. None remain
at the base of the cotyledon. There are markedly distended endometrial
blood vessels at the cotyledonary peripheries.
11)
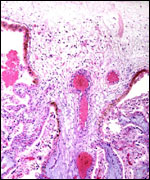
Various features
There is no subplacenta, and no metrial glands exist.
12)
Endocrinology
Melengestrol application in food has been effective in contracepting gorals
temporarily (Patton et al., 2000). Cessation of the medication reversed
the effect. I have not been able to find any literature on hormonal studies
of orals or their closer relatives.
13)
Genetics
The Chinese goral chromosome number is 2n=56 (Soma et al., 1980), all
elements beings acrocentric chromosomes. In contrast, the Japanese serow
has a diploid number of 2n=50, with 10 metacentrics, presumably derived
by Robertsonian fusion (Benirschke et al., 1972), and the Sumatran serow
with 2n=46 (Fischer & Hohn, 1972). In personal communications I learned
from Dr. Shi, L of Kunming, China, that they had examined a western Chinese
goral (N. g. griseus) with 2n=54 that had two metacentric chromosomes,
obviously derived from Robertsonian fusion of acrocentrics. This finding
helps to explain the one karyotype of 2=55 by Wurster (1972), as it may
be that that specimen represented a hybrid between animals with 2n=56
and 2n=54.
Hybrids between "Grey" and Korean gorals were reported by Gray
(1972).
14)
Immunology
I am not aware of any studies conducted in this species.
15)
Pathological features
Griner (1983) autopsied only one animal; it died with "lumpy jaws".
16)
Physiologic data
There are no published data.
17)
Other resources
Cell strains are available from CRES
at the Zoological Society of San Diego by contacting Dr. O. Ryder at:
oryder@ucsd.edu.
18) Other remarks - What additional Information is needed?
It would obviously be of interest to have some term, delivered placentas
available and specimens from related species as well. Endocrine studies
are completely lacking.
Acknowledgement
The animal photographs in this chapter come from the Zoological Society
of San Diego. I appreciate also very much the help of the pathologists
at the San Diego Zoo.
References
Benirschke, K., Soma, H. and Ito, T.: The chromosomes of the Japanese
serow, Capricornis crispus (Temminck). Proceed. Japan Acad. 48:608-612,
1972.
Dolan,
J.M.: Beitrag zur systematischen Gliederung des Tribus Rupicaprini Simpson,
1945. Z. zool. Syst. Evolutionsfschg. 1:311-407, 1963.
Fischer,
H. und Hohn, H.: Der Karyotyp der Serau (Capricornis sumatrensis,
Bechstein, 1979). Giessener Beitr. Erbpathol. Zuchthyg. 4:8-15, 1972.
Gray,
A.P.: Mammalian Hybrids. A Check-list with Bibliography. 2nd edition.
Commonwealth Agricultural Bureaux Farnham Royal, Slough, England, 1972.
Griner,
L.A.: Pathology of Zoo Animals. Zoological Society of San Diego, San Diego,
California, 1983.
Nowak,
R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press,
Baltimore, 1999.
Patton,
M.L., Aubrey, L., Edwards, M., Rieches, R., Zuba, J. and Lance, V.A.:
Successful contraception in a herd of Chinese goral (Nemorhaedus goral
arnouxianus) with melengestrol. J. Zoo Wildl. Med. 31:228-230, 2000.
Puschmann,
W.: Zootierhaltung. Vol. 2, Säugetiere. VEB Deutscher Landwirtschaftsverlag
Berlin, 1989.
Soma,
H., Kada, H., Matayoshi, K., Kiyokawa, T., Ito, T., Miyayoshi, M. and
Nagase, K.: Some chromosomal aspects of Naemorhedus goral (Goral)
and Procapra gutturosa (Mongolian Gazelle). Proceed. Japan Acad.
, Series B, 56:273-277, 1980.
Soma,
H., Kada, H., Mori, Y., Ino, O. and Hayakawa, T.: Breeding control of
serow (Capricornis) and goral (Nemorhedus) in Japan Serow
Center. Verhandl. Ber. Erkrg. Zootiere 36:49-54, 1994.
Wurster,
D.H: Sex-chromosome translocation and karyotypes in bovid tribes. Cytogenetics
11:197-207, 1972.
|