| (Clicking
on the thumbnail images will launch a new window and a larger version
of the thumbnail.) |
| Last updated: Oct 11, 2006. |
Tadarida brasiliensis
Order: Chiroptera
Family: Molossidae
1) General Zoological Data.
There are 16 genera and 86 species of Molossidae according to Nowak (1999) and they are widely distributed. Their evolutionary history is also detailed in that review. This insectivorous species (often referred to as house bat') is widely distributed throughout the Southern North American States and occurs in huge numbers in the Texan guano caves'. In the winter, most of the Tadarida brasiliensis migrate to Mexico and even further south. The animal weighs between 11 and 14 g and is frequently predated by owls, snakes, raccoons and other species, and is also a known carrier of rabies. Their longevity is 10-11 years in the wild (Weigl, 2005). Two definite subspecies are known to exist in the USA ( T.b.cynocephala & T.b.mexicana ) which are distinguished by cranial morphology and they apparently interbreed; others have suggested that there are altogether nine subspecies comprised by this species. These bats are enormously important for the destruction of large numbers of moths. Thus, their diminution in recent years due to human interference is ecologically significant. The name tadarida is debated and poorly documented; free-tail' refers to the independence of the tail form the flight membrane (Gotch, 1979).
 |
Free-tailed bat in flight (From Tuttle). |
 |
Free-tailed bat sitting (From Tuttle). |
2) General Gestational Data
The length of gestation is 77-90 days (Stephens & Easterbrook, 1969 indicated that it lasts 4 months) and singleton births are the rule. The fetuses gestate in the right uterine horn only and twins are extremely uncommon. The size of the naked neonates is about 25 mm (Hayssen, 1993). Stephens (1962) who has amassed the greatest database described it thus: Total weight of mother and fetus was 15.2g, weight of the fetus 2.7 g, and weight of the placenta 0.8 g. The placenta examined for this description weighed also less than 1 g; in fact, we were unable to determine its precise weight, lacking an appropriate scale. Van der Merwe et al. (1986) who studied reproduction in the African Tadarida pumila , concluded that breeding is seasonal, that females are polyestrous, and that gestation lasts 60 in the right uterine horn
3) Implantation
First attachment is antimesometrial and superficial placentation of this discoid, chorio-vitelline organ then takes place according to Mossman (1987). Sansom (1932), however, identified the earliest implantations in the mid-portion and lateral to the mesometrium.
4) General Characterization of the Placenta
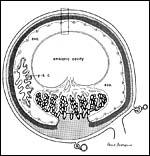
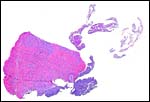
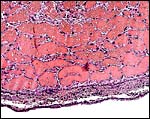
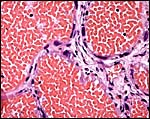
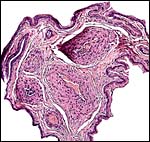
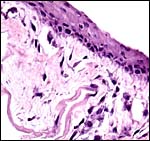
This is an endothelio-chorial placenta with areas of a labyrinthine structure and the implantation is superficial'. Mossman describes it as being hemomonochorial in type, as studied electronmicroscopically. There are no lobulations or villous formations. The maternal tubules are lined by ectoplasmic processes of trophoblast (Stephens & Cabral, 1972, as quoted by Mossman).
5) Details of fetal/maternal barrier
Details of the placentation in free-tailed bats are nearly all the result of the extensive investigations by Stephens et al. (1962 1971). Wooding & Flint (1994) illustrated (from Stephens, 1969) the electronmicroscopy of the continuous nature of the syncytiotrophoblastic attachment to the basement membrane and no direct connection to the maternal blood and Enders (1982) referred to the cellular' barrier as being hemomonochorial (results of Stephens' study). Enders et al. (1998) again referred to the central labyrinthine region of the molossid bat placenta and illustrated the maternal blood channel as being lined by (mono)-cytotrophoblast having intrasyncytial bays.
6) Umbilical cord
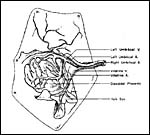
An extensive consideration of the cord structure is provided by Stephens (1962) and its sketch has been reproduced above. The cord contains 5 vessels which, according to this careful observer's description are as follows: ...a vitelline artery which comes from the dorsal aorta , a vitelline vein which enters the vena cava , a right and left umbilical artery, and the left umbilical vein. He also observed that, at term, the left umbilical vein and right umbilical artery are much larger, as can be seen in the following illustration. These are responsible for the entire blood supply to the discoidal placenta'. The left umbilical artery supplies the chorion outside of the disk.
7) Uteroplacental circulation
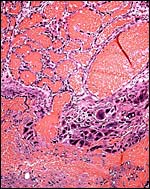
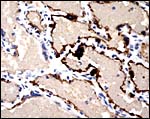
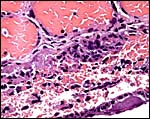
Large maternal channels are opened early in gestation around which the fetal capillary network of the labyrinth is congregated (Stephens, 1962). At this site, the endometrium degenerates, forms hemosiderin and makes giant cells. Deeper invasion by trophoblast does not take place.
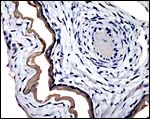
 |
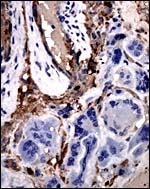
There is very irregular cytokeratin staining in the labyrinthine cells. |
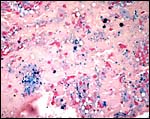 |
At the site of implantation and decidual destruction, a moderate amount of hemosiderin is found. |
8) Extraplacental membranes
There is no decidua capsularis but some decidua basalis develops and is destroyed in the superficially invasive trophoblast.
9) Trophoblast external to barrier
Stephens (1962) commented specifically that there is no deep invasion of trophoblast.
10) Endometrium
The uterus is bicornuate with the right horn being larger and the one to gestate the fetus.
Stephens (1962) has given a detailed description of the features of uterus, endometrium and associated features. Of note is the squamous nature of the endometrial lining identified by this author. While he also stated that the horns are nearly equal in size, during early gestation the right horn enlarges specifically and is nearly exclusively used for placentation. Only Sherman (1937) has once, in 300 cases, found left implantation. A decidua capsularis is not formed.
11) Various features
None remarkable features develop according to Stephens' extensive study.
12) Endocrinology
Only the right ovary is employed and usually possesses a single corpus luteum - rarely two corpora (Mossman & Duke, 1973). Stephens (1962) has speculated on the possible reasons for this feature of asymmetrical usage; he speculated that either local diffusion of progesterone from the corpus luteum or, more likely, differential blood flow can explain this. The left ovary is considerably smaller. Wimsatt (1979) has also discussed at length the asymmetry of ovarian and uterine employment for gestation in a variety of bat species; he concluded that dextral dominance is most frequent'.
13) Genetics
This species has 48 chromosomes, as displayed below. The karyotypes shown below come from Hsu & Benirschke, 1970 and they were gifts of Dr. R.J. Baker in Texas and come from a specimen collected in Oklahoma .
 |
Karyotypes of male and female free-tailed bats. |
Caire & Haydari (1990) described the absence of a distinct fetal hemoglobin. The genetic population structure was studied by McCracken et al. (1994). Hybrids between the two subspecies only are known from the wild.
14) Immunology
Transplacental rabies transmission was suggested by Ramachandran & Jayakumar (1996).
15) Pathological features
Clark et al. (1996) investigated deaths from rabies and pesticides. Similarly, Geluso et al. (1981) described pesticide residues in these bats. Other metal residues were detailed by Thies & Gregory (1994).
16) Physiologic data
Kunz & Robson (1995) reported on birth size and growth rates. The reasons for the right-sided ovulation are unknown.
17) Other resources
Numerous web sites can be accessed and these contain an enormous amount of additional information for this important species.
18) Other remarks What additional Information is needed?
More details of the distribution of trophoblast with modern staining techniques would be welcomed.
Acknowledgement
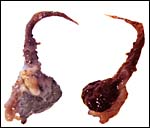
The two animal photographs in this chapter come from the website of Dr. Merlin D. Tuttle on free-tailed bats. This specimen of bat placenta was kindly made available by Drs. Deborah Kemmerer Cottrell and Manu Sebastian from Texas .
References
Caire, W. and Haydari, K.: Absence of electrophoretically distinct fetal hemoglobins in the bats Tadaria brasiliensis and Lasiurus borealis. J. Mammal. 71:695-697, 1990.
Capanna, E. and Civitelli, M.V.: Chromosomal mechanisms in the evolution of chiropteran karyotypes. Chromosomal tables of Chiroptera. Caryologia 23:79, 1970.
Clark , D.R. Jr., Lollar, A. and Cowman, D.F.: Dead and dying Brazilian free-tailed bats (Tadarida brasiliensis) from Texas : Rabies and pesticide exposure. Southwest Nat. 41:275-278, 1996.
Enders, A.C.: Whither studies of comparative placental morphology? J. Reprod. Fert. Suppl. 31:9-15, 1982.
Enders, A.C., Blankenship, T.N., Lantz, K.C. and Enders, S.S.: Morphological variation in the interhemal areas of chorioallantoic placentae. A review. Trophobl. Res. 12:1-19, 1998.
Geluso, K.J., Altenbach, J.S. and Wilson , D.E.: Organochlorine residues in young Mexican freetailed bats from several roosts. Amer. Midl. Nat. 105:249-257, 1981.
Gotch, A.F.: Mammals Their Latin Names Explained. Blandford Press, Poole , Dorset, 1979.
Hayssen, V., van Tienhoven, A. and van Tienhoven, A.: Asdell's Patterns of Mammalian Reproduction: a Compendium of Species-specific Data. Comstock/Cornell University Press, Ithaca , 1993.
Hsu, T.C. and Benirschke, K.: An Atlas of Mammalian Chromosomes. 4, Folio 159, 1970.
Kunz, T.H. and Robson, S.K.: Postnatal growth and development in the Mexican free-tailed bat (Tadarida brasiliensis mexicana): Birth size, growth rates, and age estimation. J. Mammal. 76:769-783, 1995.
McCracken, G.F., McCracken, M.K. and Vawter, A.T.: Genetic structure in migratory populations of the bat Tadarida brasiliensis mexicana . J. Mammal. 75:500-514, 1994.
Merwe, M. van der, Rautenbach, I.L. and Colf, W.J. van der: Reproduction in females of the little free-tailed bat, Tadarida (Chaerephon) pumila , in the eastern Transvaal , South Africa . J. Reprod. Fertil. 77:355-364, 1986.
Mossman, H.W.: Vertebrate Fetal Membranes. MacMillan, Houndmills, 1987.
Mossman, H.W. and Duke, K.L.: Comparative Morphology of the Mammalian Ovary. University of Wisconsin Press, Madison , Wisconsin , 1973.
Nowak, R.M.: Walker 's Mammals of the World. 6 th ed. The Johns Hopkins Press, Baltimore, 1999.
Ramachandran, S. and Jayakumar, R.: Transplacental transmission in rabies. Ind. Vet. J. 73:108-110, 1996.
Samson, G.S.: Notes on some early blastocysts of the South American bat Molossus . Proc. Zool. Soc. London, Part 1:113-118, 1932.
Sherman , H.B.: Breeding habits of the free-tailed bat. J. Mammal. 18:183-184, 1937.
Stephens, R.J.: Histology and histochemistry of the placenta and fetal membranes in the bat, Tadarida brasiliensis cynocephala (with notes on maintaining pregnant bats in captivity). Amer. J. Anat. 111:259-275, 1962.
Stephens, R.J.: The development and fine structure of the allantoic placental barrier in the bat Tadarida brasiliensis cynocephala. J. Ultrastruct. Res. 28:371-398, 1969
Stephens, R.J. and Cabral, L.: Direct contribution of the cytotrophoblast to the syncytiotrophoblast in the diffuse labyrinthine endothelio-chorial placenta of the bat. Anat. Rec. 169:243-252, 1971.
Stephens, R.J. and Cabral, L.: Cytological differentiation of the mesothelial cells of the yolk sac of the bat, Tadarida brasiliensis cynocephala. Anat. Rec. 171:293-312, 1971.
Stephens, R.J. and Cabral, L.: The diffuse labyrinthine endotheliodichorial placenta of the free-tail bat: A light and electron microscopic study. Anat Rec. 172:221-252, 1972.
Stephens, R.J. and Easterbrook, N.: A new cytoplasmic organelle, related to both lipid and glycogen storage materials in the yolk sac of the bat, Tadarida brasiliensis cynocephala . Amer. J. Anat. 124:47-56, 1969.
Stephens, R.J. and Easterbrook, N.: Development of the cytoplasmic membranous organelle in the endodermal cells of the yolk sac of the bat, Tadarida brasiliensis. J. Ultrastruct. Res. 24:239-248, 1968.
Stephens, R.J. and Easterbrook, N.: A new cytoplasmic organelle related to both lipid and glycogen storage materials in the yolk sac of the bat, Tadarida brasiliensis. Amer. J. Anat. 124:47-56, 1969.
Stephens, R.J. and Easterbrook, N.: Ultrastructural differentiation of the endodermal cells of the yolk sac of the bat, Tadarida brasiliensis. Anat. Rec. 169:207-242, 1971.
Thies, M. and McGregor, D.: Residues of lead, cadmium, and arsenic in livers of Mexican free-tailed bats. Bull. Environ. Contam. Toxicol. 52:641-648, 1994.
Weigl, R.: Longevity of Mammals in Captivity from the Living Collections of the World. E. Schweizertbart'sche Verlagsbuchhandlung (Nägele und Obermiller), Stuttgart, 2005.
Wimsatt, W.A.: Some aspects of the comparative anatomy of the mammalian placenta. Amer. J. Obstet. Gynecol. 84:1568-1594, 1962.
Wimsatt, W.A.: Reproductive asymmetry and unilateral pregnancy in Chiroptera. J. Reprod. Fertil. 56:345-357, 1979.
Wooding, F.B.P. and Flint , A.P.F.: Placentation. Chapter 4 (pp.233-460) in, G.E. Lamming, ed. Marshall's Physiology of Reproduction, 4 th ed. Vol. 3, Part 1. Chapman & Hall, London , 1994.